
Thus a lower effective nuclear charge, then flooring. Chlorine has one more energy level than flooring. Thus I'm going to call it a a tight hold putting that in quotations on electrons. So they both have very high nuclear charge effective nuclear charge. They're both on the far right of their period. And both these elements are halogen In group 78 With seven valence electrons. We are asked to account for the differences between the two elements. That's okay, so we're just going to look at all these values and compare them now based on those values. That doesn't even sound remotely close but we'll take it and 79 peak m maybe that does someone Okay, I'm going to pull this down and see what else I have here. 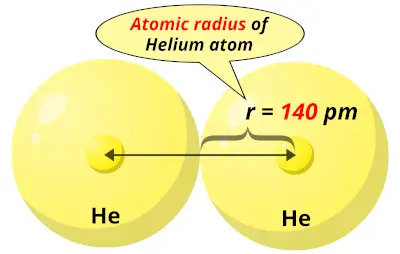
So it might not be exactly the same as the one in the text, but that's okay. Now the atomic radius I'm getting is from my my periodic table. And finally our last property is atomic radius. And for flooring It is 328 villa joules per mole. Let me get back to my periodic table again for electron affinity For Chlorine. Let's go back to blue for a letter, e letter e will be electronic affinity. Since these have such high ionization energies, they're not going to be very reactive with water. One of these is going to be terribly reactive with water reactivity with water is often based on first ionization energies being low. What haven't I used yet and let me neither. In fact, they gain electrons the reactivity towards water. Neither one of these is in a position to lose electrons. Both remember ionization energy is the energy required to remove electron from the gaseous state. Dylan, joules per mole and chlorine is 12 51 killer jewels per mole. I'm going to find flooring by looking at my periodic table. So hang on should have probably done that. Our next item is first ionization energy. Bye ending up with a to S to two P 6 or a three S two, three P six shell or outer configuration. Each of these wants to achieve a noble gas configuration. Our second item is the most common charge for the ion. So look at the last piece of each and we'll see that the electron configurations for the highest filled energy level is the same two s. So, I'm going to write the electron configuration for each And we have one s 2, 2 S two. The first we are asked to look at is electron configuration. We are asked to make several comparisons for different properties between chlorine and chlorine. Mean is, uh, Rhonda won't white one for extra unhand. Mean he's created under for curing because bro be in its MP Got hot sun and coming Prettiest girl for Rome. Create IUs have got something created us. We are asked curing gas with the actress water Jim Prentice HCL loss each hole C l Then the next one is uh, um tell me. So bro me where the actress watcher us, uh, gas be out your gas to produce Be, huh? HB How on da hates whole be har. Then we have to compare, uh, reactivity towards water. First he a nice Asian energy off Dramamine thing. Then the next one is the first ionization energy. So most common dining challenge for Romanians minus one for Korean e. Look at the most call on high ionic charges. 
And Romeo will be gone for lest you fall p by then be he's, uh we need thio. So you can just say the electric off Gorshin off chlorine will be neon neon, followed by two Yes, two and three p five. Okay, so we have Ah, bro, Ming is electron configuration sobral mean And, uh, chlorine stands with chlorine puree. So we have ah remain including and were sent to compere Ah, the electron configuration. The next question say's computer the element bromine and chlorine with respect to the falling property.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
